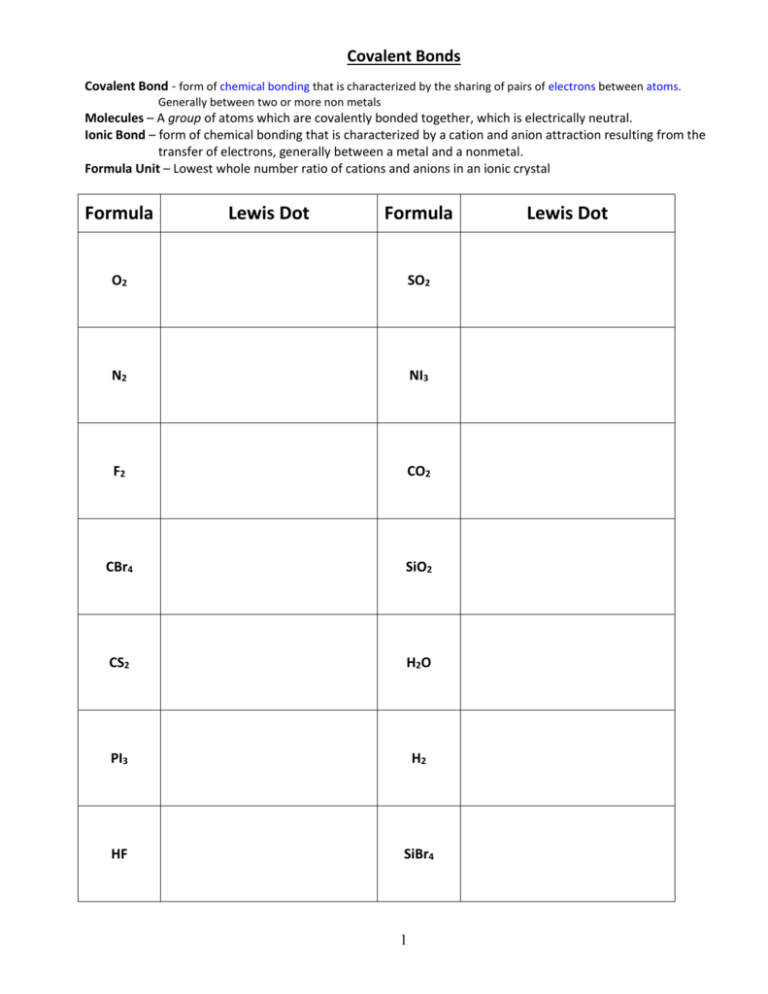
SF2 Bond Angle Whenever the canter atom has two lone pairs and two particles, the geometry is bent or angular. Resonance structures can also be non-equivalent, in which case they will have different numbers and/or locations of bonds. The molecule SO2 (shown above) has two such resonance forms. Examples are shown for the molecules SF2 and CH2O below. Pairs of dots are used to represent lone pair electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
